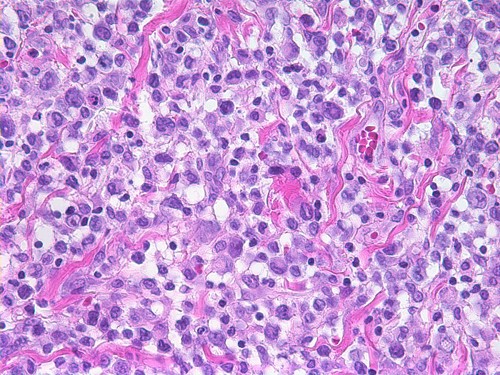
Breast Implant Associated Anaplastic Large Cell Lymphoma (BIA-ALCL)
Since the last BIA-ALCL update, all the plastic surgery societies and breast implant manufacturers have responded with information sheets. I am encouraged by the free flow of information within the Plastic Surgery profession and the industry that supports us.
ASAPS by the Numbers
The ASAPS has distributed a quick summary of the numbers related to BIA-ALCL. Here are a few.
How Many BIA-ALCL Cases Have Been reported?
Of the estimated 7 million women with breast implant throughout the world, 359 reports have been filed. Since the reports can be filed by patients, physicians or manufacturers, it’s important to note the MDRs (Medical Device Reports) are not individual cases, and multiple reports may be filed for a single case of BIA-ALCL. In contrast, there are currently 139 case reports of BIA-ALCL in the literature. It is also important to realize that this is a very small number in relation to the millions of breast implant patients. Lastly, the number is expected to rise because we are learning how better to identify and diagnose patients with the disease.
Smooth vs. Textured
Of the reported cases, over 90% occurred in patients who had textured implants at the time of their diagnosis. In the US database, there is a single case of a patient with smooth breast implants developing BI-ALCL; however, they had a textured tissue expander prior to the smooth breast implant placement. Unfortunately, the other 27 cases of BIA-ALCL reported in the world databases do not include past breast implant information, so while we can say that BI-ALCL is predominantly a problem with textured breast implants, it is unknown if a case has developed in a person who had exclusively smooth breast implants. Currently, 12.7% of US patients receive textured implants. This includes both textured round and all anatomically “shaped” breast implants, and includes both saline filled and silicone filled breast implants.
How Many BIA-ALCL Deaths Have Been Reported?
Of the 359 cases reported, 9 patients have died. Two patients died from stem cell transplants, one died from development of a second unrelated lymphoma, and 6 patients died from direct extension of the tumor. Early detection is key. Of the deaths, none of them received complete surgical excision at any point in their clinical history, none received targeted therapy, and most were significantly delayed in diagnosis or receiving any treatment (1-2 years from onset of symptoms). On the other hand, 93% of all patients are disease free at 3 years follow up. BIA-ALCL has an excellent prognosis when diagnosed early.
How Do We Diagnose BIA-ALCL?
Early diagnosis is key to successful treatment, so how do we accomplish this? Unilateral (one-sided) swelling or mass should lead to a thorough evaluation. This is common in the months following your breast augmentation. The possibility of BIA-ALCL should be considered in a patient with late onset, peri-implant seroma (fluid collection). The majority of patients present on average 8 years after implantation, but range extends from 2 to 28 years.
Ultrasound imaging followed by fine needle aspiration of seroma fluid should be obtained. Collected fresh seroma fluid should be sent to a qualified lab for immunohistochemistry testing for cluster of differentiation (CD30) testing. Anaplastic Lymphocyte Kinase (ALK) testing of the seroma fluid can help differentiate BIA-ALCL from systemic forms of lymphoma that may appear in the breast.
Current BIA-ALCL Treatment
Most patients with BIA-ALCL are cured by removal of the implant and capsule surrounding the implant, however a small number act more aggressively and may require additional treatments. For 2017, the National Comprehensive Cancer Network defines optimal treatment which is total capsulectomy and implant removal for the majority of patients with disease confined to the capsule (35% of patients) or a resectable mass (40% of patients). Advanced disease with lymph node metastasis (14% of patients) or organ metastasis (1% of patients) may require further treatment with chemotherapy using either CHOP anthracycline based-protocol or targeted therapy with brentuximab vedotin. Radiation therapy is only reserved for locally unresectable disease.
BIA-ALCL Perspective
BI-ALCL is not breast cancer. Here are some additional numbers, and some links, to keep the worry about BIA-ALCL in perspective:
- The average woman’s risk of developing breast cancer is 12.5%.
- The risk of developing recurrent breast cancer after mastectomy is 5-8%.
- The risk of capsular contracture through 10 years is 14%.
- The risk of breast implant rupture through ten years is 10%.
- The risk of developing BIA-ALCL is 0.003%
- Understanding Rare Adverse Sequelae of Breast Implants
- NCCN Consensus Guidelines for the Diagnosis and Management of BIA-ALCL