Top 10 Breast Augmentation Frequently Asked Questions
Today, we review the Top 10 Frequently Asked Questions about Breast Augmentation. These questions come from my patients and from inquiries on sites like […]
Top 10 Mommy Makeover Frequently Asked Questions (FAQs)
The Mommy Makeover remains one of the most commonly performed Cosmetic Plastic Surgery Procedure combinations in the San Francisco Bay Area. Today, on the […]
Breast Implants 101 What You Need to Know
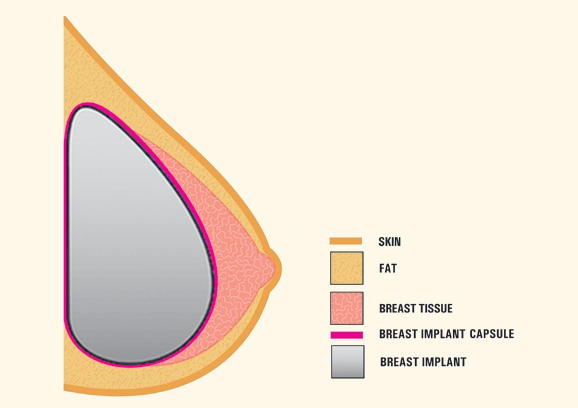
Breast Implants are medical devices designed to enhance the size, shape, and overall appearance of the breasts. They are widely used for cosmetic reasons, […]
Statement on Breast Implant Capsulectomy Definitions 2024
Surgical treatment of breast implant capsules are varied and diverse. Today, I want to review the available options and outline when they are used. […]
Downsizing Breast Implants – What You Need to Know
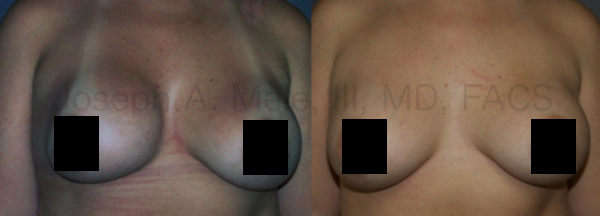
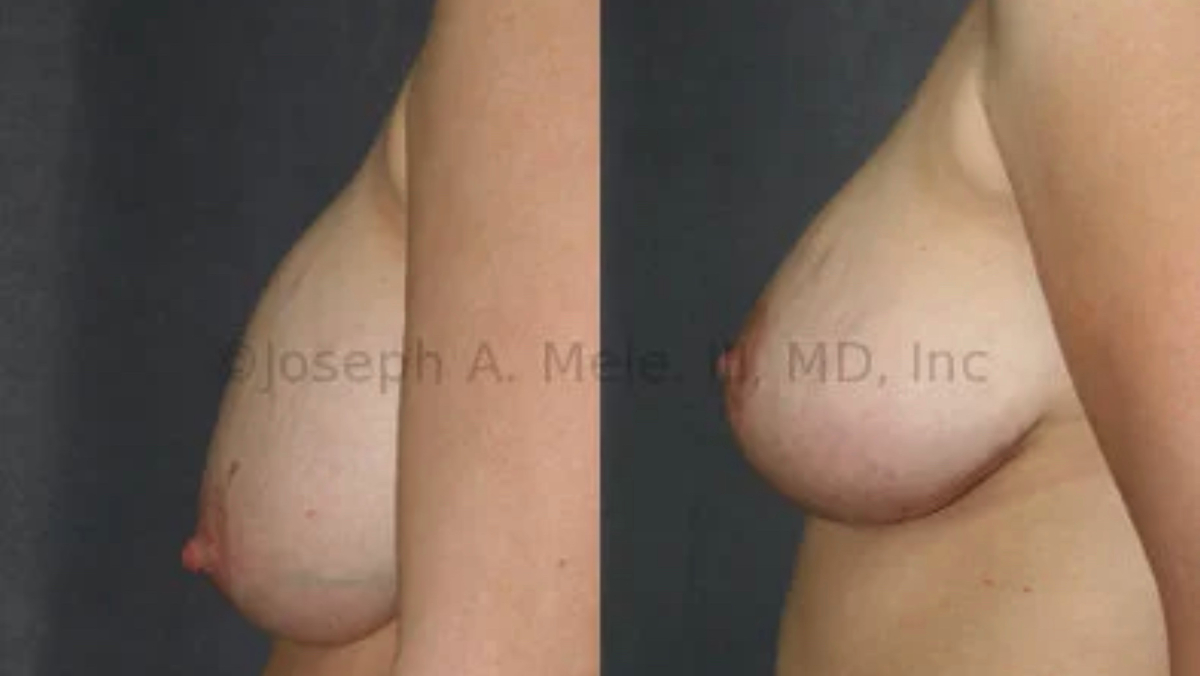
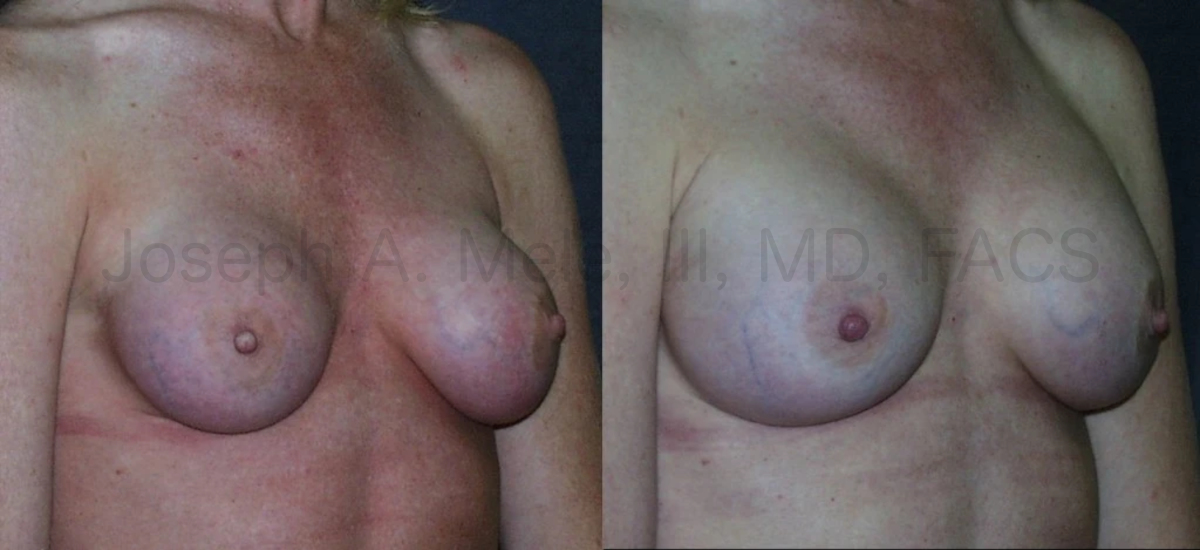
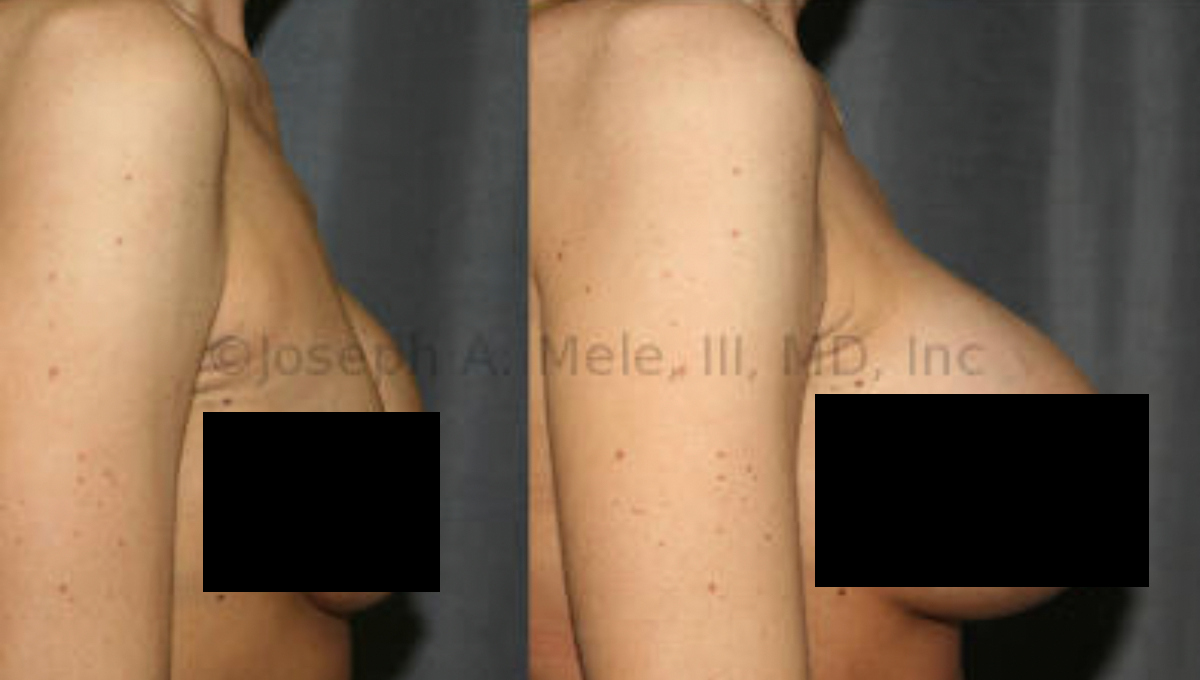
Breast Augmentation remains the most popular elective cosmetic plastic surgery procedure and has one of the highest patient satisfaction ratings. The ability to provide […]
Breast Implant Maintenance and Resizing
Breast Augmentation remains the most popular cosmetic plastic surgery. Whether placed for reconstructive or cosmetic reasons, Breast Implant Revision is needed from time to […]
Breast Cancer and Breast Implant Associated Cancer Update
In the United States there are about 270,000 cases of Breast Cancer each year, and 99% of them are in women. If you are […]
Breast Implant Maintenance and Breast Revision Surgery
Breast Augmentation Revision is used to maintain the aesthetic results of breast enhancing surgery. As we discussed last week, Breast Implants are not lifetime […]
Breast Implant Maintenance and Breast Revision Surgery
Breast Implants have come a long way over their 60 years of existence. They are available in a wider range of shapes, volumes and […]
Breast Implants and Mammograms
I subscribe to many feeds that feature the greatest and latest plastic surgery news. Much of it is smoke and mirrors, but every now […]