Dermal Fillers for Facial Wrinkle Reduction – Walnut Creek Liquid Facelift
The Liquid Facelift is a quick way to reduce the signs of facial aging. Technically not a facelift, the term liquid facelift refers to […]
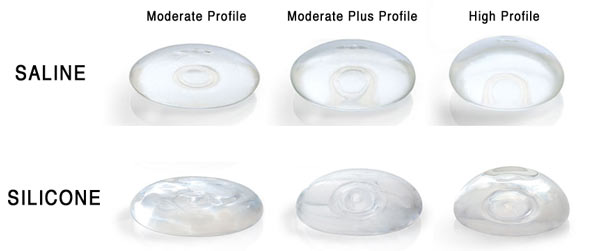
Silicone Breast Implants Turn 50
As 2011 draws to a close, so do the first 50 years of breast implants. In 1961, American plastic surgeons Thomas Cronin and Frank […]
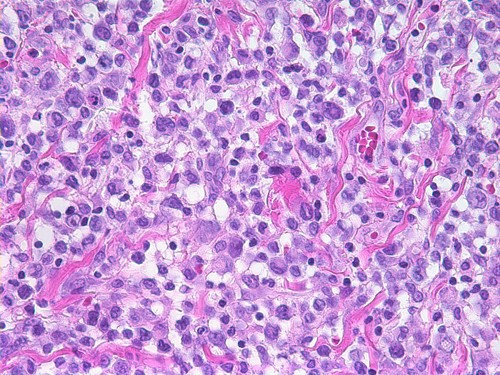
ALCL Update – Breast Implants and Anaplastic Large Cell Lymphoma Update
Earlier this year, the San Francisco Plastic Surgery Blog reported an extremely unusual, but possible association between breast implants and a rare form of […]